Alrich Lawson
In 1972, Janet Rowley sat at her dining room table and cut out tiny chromosomes from photographs she had taken in her lab. She cut out each one of the tiny dolls her children teasingly called “paper dolls.” Then she carefully arranged them in 23 pairs, warning her children not to sneeze on them.
The physician-scientist had just mastered new chromosome-staining techniques during a year-long sabbatical from Oxford University, but it was in the dining room of her Chicago home that she made a discovery that would dramatically change the course of cancer research.
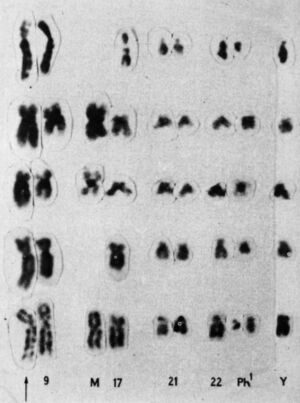
While examining the chromosomes of patients with acute myeloid leukemia (AML), Chromosomes 8 and 21 had been broken and swapped positions.This is a genetic swap called a translocation, and when she looked at the chromosomes of other patients with AML, she found the same swap – the 8;21 translocation.
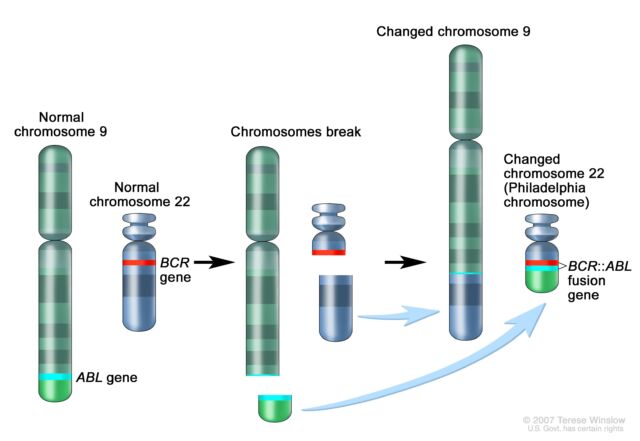
Later that year, she saw another translocation, this time in a patient with a different type of blood cancer called chronic myeloid leukemia (CML). CML patients were known to have a mysterious abnormality in chromosome 22 that makes it appear shorter than normal. This abnormality Philadelphia chromosome Two people In 1959, a researcher from PhiladelphiaBut it wasn’t until Lawrie took a closer look at her carefully set dining table that the reason for chromosome 22’s shortness became apparent: part of it had broken off and replaced a small section of chromosome 9 – a 9:22 translocation.
Lowry had the first evidence that genetic abnormalities cause cancer. She published her findings in 1973, and said that CML translocations Single authored study in NatureOver the next few years, she pushed the idea that abnormalities were important to cancer, but initially met with skepticism. At the time, most researchers believed that chromosomal abnormalities were the result of cancer, not the other way around. Lowry’s results were rejected by the prestigious New England Journal of Medicine. “At first, it was met with a kind of amusement and tolerance,” she says. Before her death in 2013, she said:.
The birth of targeted therapy
But the evidence was mounting quickly: in 1977, Lawrie and two of her colleagues at the University of Chicago identified another chromosomal translocation.15;17This causes a rare blood cancer called acute promyelocytic leukemia. By 1990, more than 70 translocations in cancer had been identified.
Its importance also grew rapidly. After Lawry discovered the 9;22 translocation in CML, researchers discovered that a genetic swap results in the fusion of two genes. Part of the ABL gene, normally found on chromosome 9, attaches to the BCR gene on chromosome 22, creating the cancer-causing BCR::ABL fusion gene on chromosome 22. This gene fusion codes for a signaling protein (tyrosine kinase) that remains in a permanently “active” mode. This permanently triggers a signaling pathway that causes white blood cells to proliferate uncontrollably.
By the mid-1990s, researchers developed drugs called tyrosine kinase inhibitors (TKIs) that block the BCR-ABL protein. ImatinibFor patients in chronic phase CML (approximately 90% of CML patients), imatinib increases 10-year survival rates from less than 50% to Just over 80 percentImatinib (sold as Gleevec or Gleevec) received approval from the Food and Drug Administration in 2001 and was the first cancer treatment approved to target a known genetic mutation.
The success of imatinib marked the beginning of targeted cancer treatment, or precision medicine. By the early 2000s, researchers were increasingly interested in pinpointing the genetic basis of cancer. At the same time, revolutionary developments in next-generation gene sequencing served as jet fuel for this burgeoning field. This technology made it easier to identify the mutations and genetic abnormalities that cause cancer. Sequencing is now considered the standard of care in the diagnosis, treatment, and management of many cancers.
The development of gene-targeted cancer therapy has progressed dramatically, and the TKI class, such as imatinib, has expanded particularly rapidly. Over 50 FDA-approved TKIs They target a variety of cancers. For example, TKIs such as lapatinib, neratinib, tucatinib, and pyrotinib, Human epidermal growth factor receptor 2 (HER2Janus kinase 2 (K2) is a kinase that is frequently mutated in rare blood cancers. Myelofibrosis And that Polycythemia vera, a slow-growing blood cancerMeanwhile, CML patients currently have five TKI therapies to choose from.



